Proposal & Approach
From 2006.igem.org
| Line 15: | Line 15: | ||
<ol> | <ol> | ||
<li>New mechanism of tolerance | <li>New mechanism of tolerance | ||
| - | - use of inhibitors that interfere with NFκB (transcription factor mentioned above) | + | - use of inhibitors that interfere with [[Troubleshooting, References & Sponsors#Terms|NFκB]] (transcription factor mentioned above) |
- inhibition with dominant negative proteins involved in signaling pathway, this proteins could be labeled with degradation tags (PEST sequence) and inhibition would be temporal (negative feedback loop)</li> | - inhibition with dominant negative proteins involved in signaling pathway, this proteins could be labeled with degradation tags (PEST sequence) and inhibition would be temporal (negative feedback loop)</li> | ||
| Line 27: | Line 27: | ||
<p>The basic idea of our project was to introduce the feedback loop,which would decrease the response to the persistent or repeated stimulus. However completly shutting down the response at bacterial stimulation is not a good solution. Ideally the feedback loop should decrease the response when it is too high but recover the responsiveness of the system after some time.</p> | <p>The basic idea of our project was to introduce the feedback loop,which would decrease the response to the persistent or repeated stimulus. However completly shutting down the response at bacterial stimulation is not a good solution. Ideally the feedback loop should decrease the response when it is too high but recover the responsiveness of the system after some time.</p> | ||
| - | <p>Inhibition of the response could be achieved by activating the expression of the dominant-negative adapter protein, that inactivates the signaling pathway. Decreasing the lifetime of the dominant-negative inhibitior by the addition of rapid degradation tag (PEST sequence) should inactivate the inhibition and reset (restore) the normal responsiveness of the immune system.</p> | + | <p>Inhibition of the response could be achieved by activating the expression of the [[Troubleshooting, References & Sponsors#Terms|dominant-negative]] adapter protein, that inactivates the signaling pathway. Decreasing the lifetime of the dominant-negative inhibitior by the addition of rapid degradation tag ([[Troubleshooting, References & Sponsors#Terms|PEST sequence]]) should inactivate the inhibition and reset (restore) the normal responsiveness of the immune system.</p> |
<p>This idea is similar to the natural mechanism of tolerance, which is already present in living cells and which decrease the response to repeated bacterial stimulation. This natural tolerance is activated slowly, on the order of days and operates through several different mechanisms (Figure). Our feedback mechanism (i.e. artificial tolerance) should decrease the response within hours and thus "attack" the signaling pathway at the point, which has not been used in the natural system.</p> | <p>This idea is similar to the natural mechanism of tolerance, which is already present in living cells and which decrease the response to repeated bacterial stimulation. This natural tolerance is activated slowly, on the order of days and operates through several different mechanisms (Figure). Our feedback mechanism (i.e. artificial tolerance) should decrease the response within hours and thus "attack" the signaling pathway at the point, which has not been used in the natural system.</p> | ||
| Line 57: | Line 57: | ||
<p>At first we had to design primers to replicate a desired DNA fragment. In primers we included restriction sites - on left site XbaI and on the right site SpeI, NcoI and PstI. We cloned that part in to BioBrick plasmids with ccdB domain to get all restriction sites needed for BioBrick assembly. We had to design all parts de novo, since no parts like promoters, terminators, desired proteins for signaling pathway modification, degradation flags and reporters had been designed so far - neither to work in mammalian cells. List of desired constructs is shown below. For our use we designed a special vector ([http://partsregistry.org/Part:BBa_J52017 BBa_J52017]) with terminator to simplify constructs assembly. All our composite parts (promoter plus part) were then cloned in this vector.</p> | <p>At first we had to design primers to replicate a desired DNA fragment. In primers we included restriction sites - on left site XbaI and on the right site SpeI, NcoI and PstI. We cloned that part in to BioBrick plasmids with ccdB domain to get all restriction sites needed for BioBrick assembly. We had to design all parts de novo, since no parts like promoters, terminators, desired proteins for signaling pathway modification, degradation flags and reporters had been designed so far - neither to work in mammalian cells. List of desired constructs is shown below. For our use we designed a special vector ([http://partsregistry.org/Part:BBa_J52017 BBa_J52017]) with terminator to simplify constructs assembly. All our composite parts (promoter plus part) were then cloned in this vector.</p> | ||
| - | <p>We also needed fusion proteins e.g. dnMyd88-rLuc-PEST ([http://partsregistry.org/Part:BBa_J52013 BBa_J52013]) - that is our dominant negative protein linked with reporter and degradation flag. This parts are designed like basic parts - not composite, although they are fusion proteins. Between proteins there is a 6 aminoacid long linker because only 2 aminoacid long linker formed during biobrick assembly could affect protein folding. We introduced a six amino acids long linker in between protein - reporter and reporter - degradation flag with primers using PCR Overlap Extension method. These parts were then combined with promoter (NFκB) in BioBrick assembly technique. The part was then inserted in vector with terminator and ready to use in human cells (HEK 293).</p> | + | <p>We also needed fusion proteins e.g. dnMyd88-rLuc-PEST ([http://partsregistry.org/Part:BBa_J52013 BBa_J52013]) - that is our dominant negative protein linked with reporter and degradation flag. This parts are designed like basic parts - not composite, although they are fusion proteins. Between proteins there is a 6 aminoacid long linker because only 2 aminoacid long linker formed during biobrick assembly could affect protein folding. We introduced a six amino acids long linker in between protein - reporter and reporter - degradation flag with primers using PCR Overlap Extension method. These parts were then combined with promoter ([[Troubleshooting, References & Sponsors#Terms|NFκB]]) in BioBrick assembly technique. The part was then inserted in vector with terminator and ready to use in human cells (HEK 293).</p> |
<center><table> | <center><table> | ||
| Line 119: | Line 119: | ||
<h2>Transfection</h2> | <h2>Transfection</h2> | ||
| - | <p>In September and October we started to work on transfection of our constructs into human embrional kidney cells (HEK293). and three detection system mentioned below. At first we had to optimize the methods (read articles, test negative and positive controls) and learn how to work with human cells. Experiments are still in progress.</p> | + | <p>In September and October we started to work on transfection of our constructs into human embrional kidney cells ([[Troubleshooting, References & Sponsors#Terms|HEK293]]). and three detection system mentioned below. At first we had to optimize the methods (read articles, test negative and positive controls) and learn how to work with human cells. Experiments are still in progress.</p> |
| - | <p>Despite transfection with our construct, we also have to transfect cellc with TLRs, because strain HEK293 expresses only TLR3 and TLR6. Sepsis is usually response to pathogenic Gramm negative bacteria. Their outer membrane contains LPS (lipoproteins) that is recognized by TLR4 and accessory molecule MD2. At the beginning we transformed all cells with our constructs and additional plasmids, one coding TLR4 receptor and another MD2. Very soon we found out that our plasmids were contaminated with LPS of ''E. coli'' (strain DH5α used for transformation), since there was no difference in response between stimulated (with LPS) and unstimulated cells. To overcome this problem, now we are using TLR5 receptor. This receptor detects bacterial flagelin. Signal transfer through this receptor does not depend on presence of LPS.</p> | + | <p>Despite transfection with our construct, we also have to transfect cellc with TLRs, because strain [[Troubleshooting, References & Sponsors#Terms|HEK293]] expresses only [[Troubleshooting, References & Sponsors#Terms|TLR3]] and [[Troubleshooting, References & Sponsors#Terms|TLR6]]. Sepsis is usually response to pathogenic Gramm negative bacteria. Their outer membrane contains [[Troubleshooting, References & Sponsors#Terms|LPS]] (lipoproteins) that is recognized by TLR4 and accessory molecule [[Troubleshooting, References & Sponsors#Terms|MD2]]. At the beginning we transformed all cells with our constructs and additional plasmids, one coding [[Troubleshooting, References & Sponsors#Terms|TLR4]] receptor and another [[Troubleshooting, References & Sponsors#Terms|MD2]]. Very soon we found out that our plasmids were contaminated with [[Troubleshooting, References & Sponsors#Terms|LPS]] of ''E. coli'' (strain DH5α used for transformation), since there was no difference in response between stimulated (with [[Troubleshooting, References & Sponsors#Terms|LPS]]) and unstimulated cells. To overcome this problem, now we are using [[Troubleshooting, References & Sponsors#Terms|TLR5]] receptor. This receptor detects bacterial [[Troubleshooting, References & Sponsors#Terms|flagelin]]. Signal transfer through this receptor does not depend on presence of LPS.</p> |
<h2>Detection systems</h2> | <h2>Detection systems</h2> | ||
| - | <p>We have tested our hypothesis using three different detection systems. All of them had different approach, however we were expecting the same result – synthesis of dominant negative protein (MyD88 or TRAF6), blocking of signalling pathway and consequently transcription termination of dominant negative protein. The construct (parts) we have made are designed for each detection system respectively.</p> | + | <p>We have tested our hypothesis using three different detection systems. All of them had different approach, however we were expecting the same result – synthesis of dominant negative protein ([[Troubleshooting, References & Sponsors#Terms|MyD88]] or [[Troubleshooting, References & Sponsors#Terms|TRAF6]]), blocking of signalling pathway and consequently transcription termination of dominant negative protein. The construct (parts) we have made are designed for each detection system respectively.</p> |
<p>Requests for the optimal detection system were: | <p>Requests for the optimal detection system were: | ||
| Line 148: | Line 148: | ||
Used composite parts: NFκB-MyD-Luc ([http://partsregistry.org/Part:BBa_J52014 BBa_J52014]). | Used composite parts: NFκB-MyD-Luc ([http://partsregistry.org/Part:BBa_J52014 BBa_J52014]). | ||
| - | <p>This is a method for very precise detection of each individual cell which is marked. In our case markers were secondary antibodies against phosphorilated ERK kinases. A result of transfection with dnMyD88 or dnMyD88-rLuc and stimulation with flagelin or LPS (depends on chosen TLR receptor) there was activation of signalling pathway and phosphorilation of ERK kinases. After synthesis dominant negative protein should block signalling pathway and amount of phosphorilated kinases should decrease. Further the fluorescence of fluorochromes, conjugated on secondary antibody, would decrease too.</p> | + | <p>This is a method for very precise detection of each individual cell which is marked. In our case markers were secondary antibodies against phosphorilated [[Troubleshooting, References & Sponsors#Terms|ERK kinases]]. A result of transfection with dnMyD88 or dnMyD88-rLuc and stimulation with flagelin or [[Troubleshooting, References & Sponsors#Terms|LPS]] (depends on chosen TLR receptor) there was activation of signalling pathway and phosphorilation of [[Troubleshooting, References & Sponsors#Terms|ERK kinases]]. After synthesis [[Troubleshooting, References & Sponsors#Terms|dominant negative protein]] should block signalling pathway and amount of phosphorilated kinases should decrease. Further the fluorescence of fluorochromes, conjugated on secondary antibody, would decrease too.</p> |
| Line 155: | Line 155: | ||
Used composite parts: NFκB-MyD-luc ([http://partsregistry.org/Part:BBa_J52014 BBa_J52014]), NFκB-MyD-luc-PEST([http://partsregistry.org/Part:BBa_J52024 BBa_J52024]), NFκB-luc-PEST([http://partsregistry.org/Part:BBa_J52023 BBa_J52023]), CMV-luc([http://partsregistry.org/Part:BBa_J52038 BBa_J52038]) and CMV-luc-PEST([http://partsregistry.org/Part:BBa_J52039 BBa_J52039]). | Used composite parts: NFκB-MyD-luc ([http://partsregistry.org/Part:BBa_J52014 BBa_J52014]), NFκB-MyD-luc-PEST([http://partsregistry.org/Part:BBa_J52024 BBa_J52024]), NFκB-luc-PEST([http://partsregistry.org/Part:BBa_J52023 BBa_J52023]), CMV-luc([http://partsregistry.org/Part:BBa_J52038 BBa_J52038]) and CMV-luc-PEST([http://partsregistry.org/Part:BBa_J52039 BBa_J52039]). | ||
| - | <p>With parts that are under NFκB promoter we want to prove inhibitory effect of dnMyD88 on signal pathway (on the basis of luminiscence decrease). Parts with PEST tag should be degradated and therefor luminiscence should increase. We found out that it takes 4 hours for any response.</p> | + | <p>With parts that are under [[Troubleshooting, References & Sponsors#Terms|NFκB]] promoter we want to prove inhibitory effect of dnMyD88 on signal pathway (on the basis of luminiscence decrease). Parts with [[Troubleshooting, References & Sponsors#Terms|PEST]] tag should be degradated and therefor luminiscence should increase. We found out that it takes 4 hours for any response.</p> |
| - | <p>We also want to calculate half-life of rLuc and rLuc-PEST, so we used those two constructs that are expressed under constituitive promoter CMV. Synthesis was inhibited by adding cycloheximide in intervals.</p> | + | <p>We also want to calculate half-life of rLuc and rLuc-PEST, so we used those two constructs that are expressed under constituitive promoter [[Troubleshooting, References & Sponsors#Terms|CMV. Synthesis was inhibited by adding cycloheximide in intervals.</p> |
| Line 162: | Line 162: | ||
Used composite parts: NFκB–dnMyD88 ([http://partsregistry.org/Part:BBa_J52036 BBa_J52036]) and NFκB–TRAF6–GFP ([http://partsregistry.org/Part:BBa_J52022 BBa_J52022]). | Used composite parts: NFκB–dnMyD88 ([http://partsregistry.org/Part:BBa_J52036 BBa_J52036]) and NFκB–TRAF6–GFP ([http://partsregistry.org/Part:BBa_J52022 BBa_J52022]). | ||
| - | <p>With this detection system we are detecting active NF-kappaB tranctiption factor. Active form of this protein is present after activation of signalling pathway when protein is released from complex of inhibitors (IKKα, IKKβ, IKKγ). It migrates into nucleus where acts as transcription activator. We are using high bind 96 well plates coated with streptavidine (we prepare it by ourselfes). The biotinilated probe has double stranded NFκB binding sequence and single stranded linkage with a plate. The NFκB from whole cell lysate is captured by a probe. We detect it with primary mouse antibodys and secondary peroxidase-conjugated rabbit anti-mouse antibodies.</p> | + | <p>With this detection system we are detecting active NF-kappaB tranctiption factor. Active form of this protein is present after activation of signalling pathway when protein is released from complex of inhibitors (IKKα, IKKβ, IKKγ). It migrates into nucleus where acts as transcription activator. We are using high bind 96 well plates coated with [[Troubleshooting, References & Sponsors#Terms|streptavidine]] (we prepare it by ourselfes). The biotinilated probe has double stranded [[Troubleshooting, References & Sponsors#Terms|NFκB]] binding sequence and single stranded linkage with a plate. The [[Troubleshooting, References & Sponsors#Terms|NFκB]] from whole cell lysate is captured by a probe. We detect it with primary mouse antibodys and secondary peroxidase-conjugated rabbit anti-mouse antibodies.</p> |
Revision as of 07:23, 23 October 2006

| Home | Background and Signalling Pathway | Anticipated Results & Significance | Troubleshooting, References & Sponsors | Team members |
|---|
Contents |
Project proposal
In the beginning we have discussed following project ideas:
- New mechanism of tolerance - use of inhibitors that interfere with NFκB (transcription factor mentioned above) - inhibition with dominant negative proteins involved in signaling pathway, this proteins could be labeled with degradation tags (PEST sequence) and inhibition would be temporal (negative feedback loop)
- Cell response to pathogen, that cells usualy can not recognise - for example response to beta glucans of fungi
- Find a connection/shortening of signaling pathways to make it more efficient and include more responses
Selected project proposal
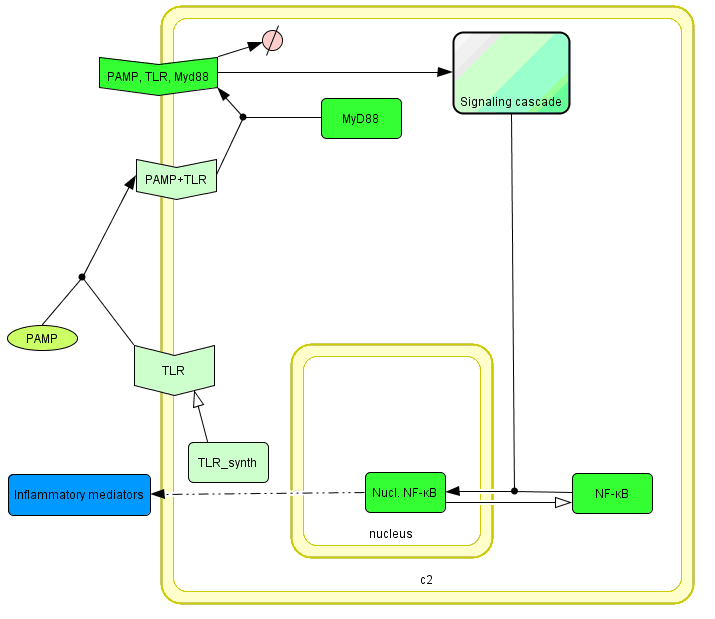
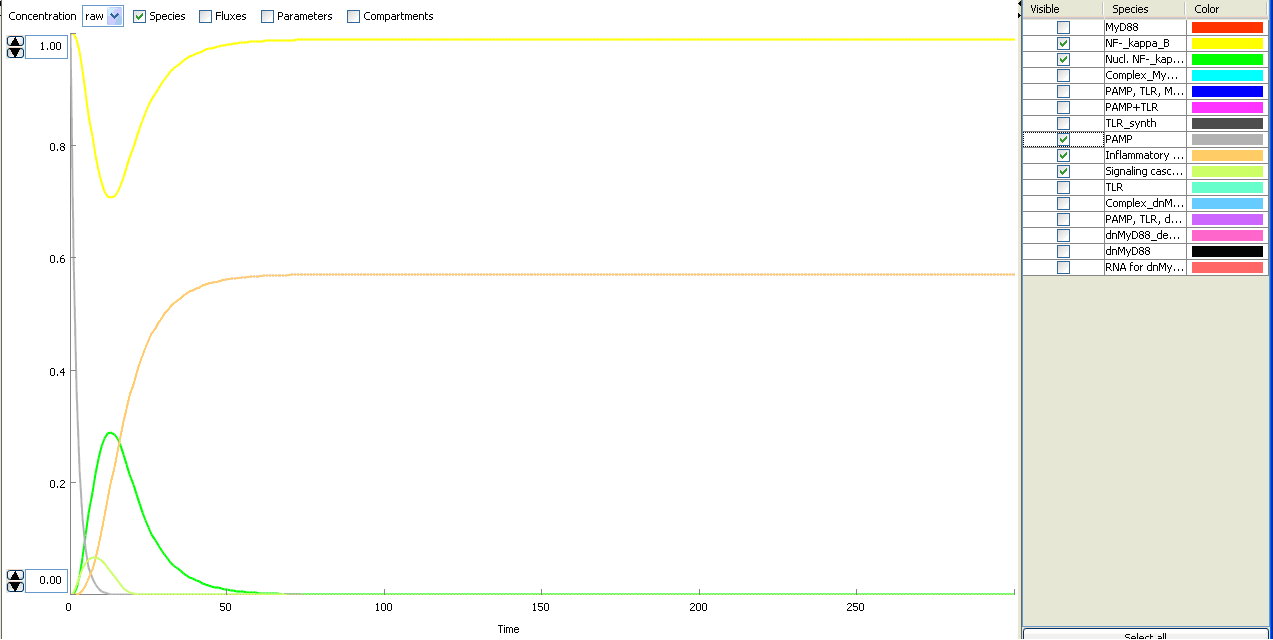
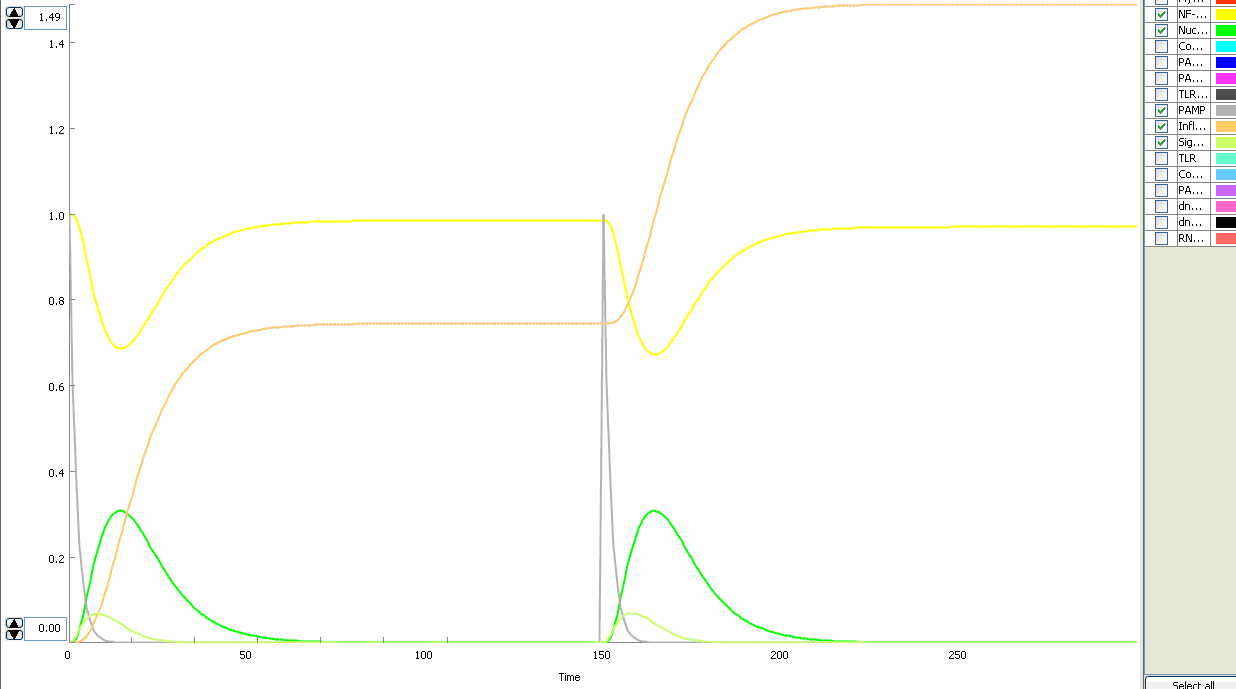
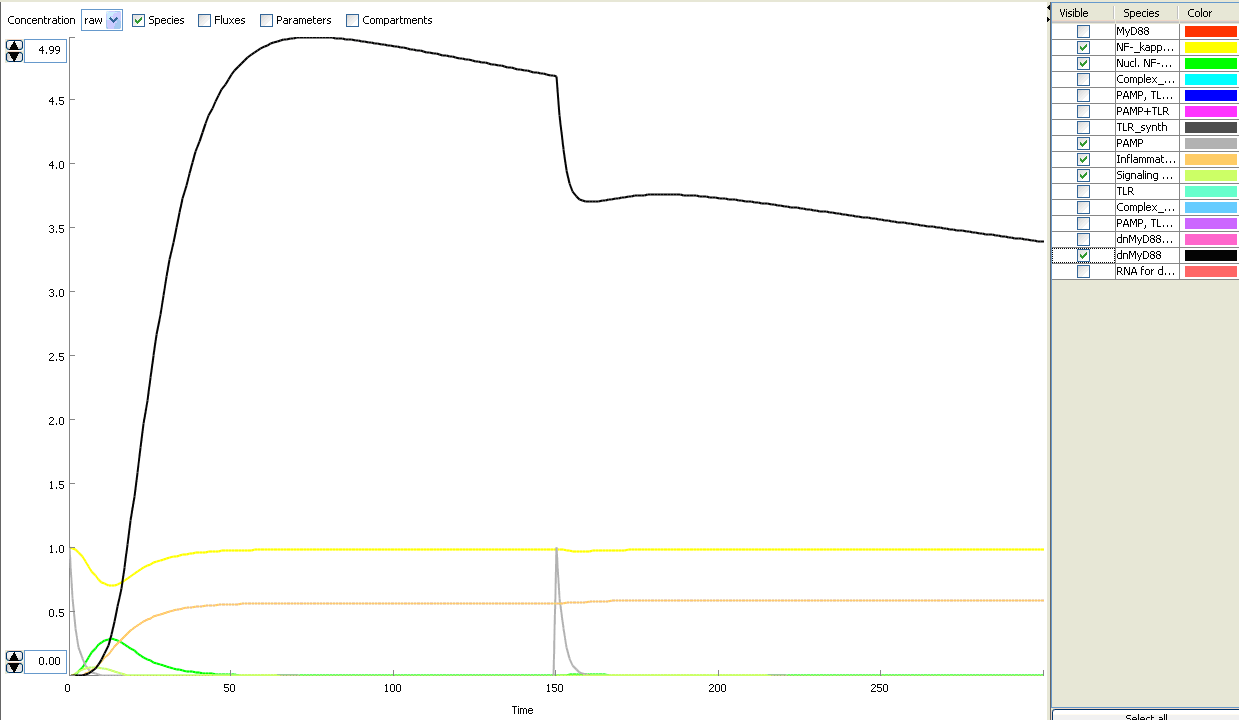
The basic idea of our project was to introduce the feedback loop,which would decrease the response to the persistent or repeated stimulus. However completly shutting down the response at bacterial stimulation is not a good solution. Ideally the feedback loop should decrease the response when it is too high but recover the responsiveness of the system after some time.
Inhibition of the response could be achieved by activating the expression of the dominant-negative adapter protein, that inactivates the signaling pathway. Decreasing the lifetime of the dominant-negative inhibitior by the addition of rapid degradation tag (PEST sequence) should inactivate the inhibition and reset (restore) the normal responsiveness of the immune system.
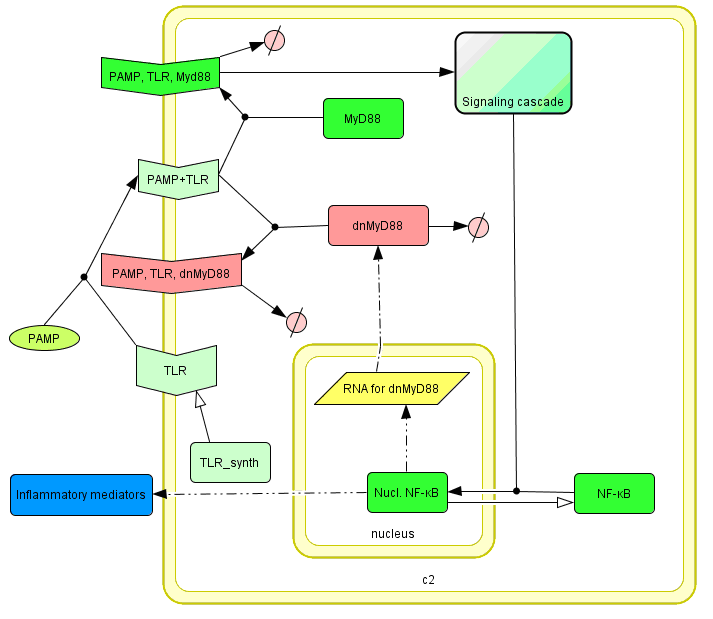
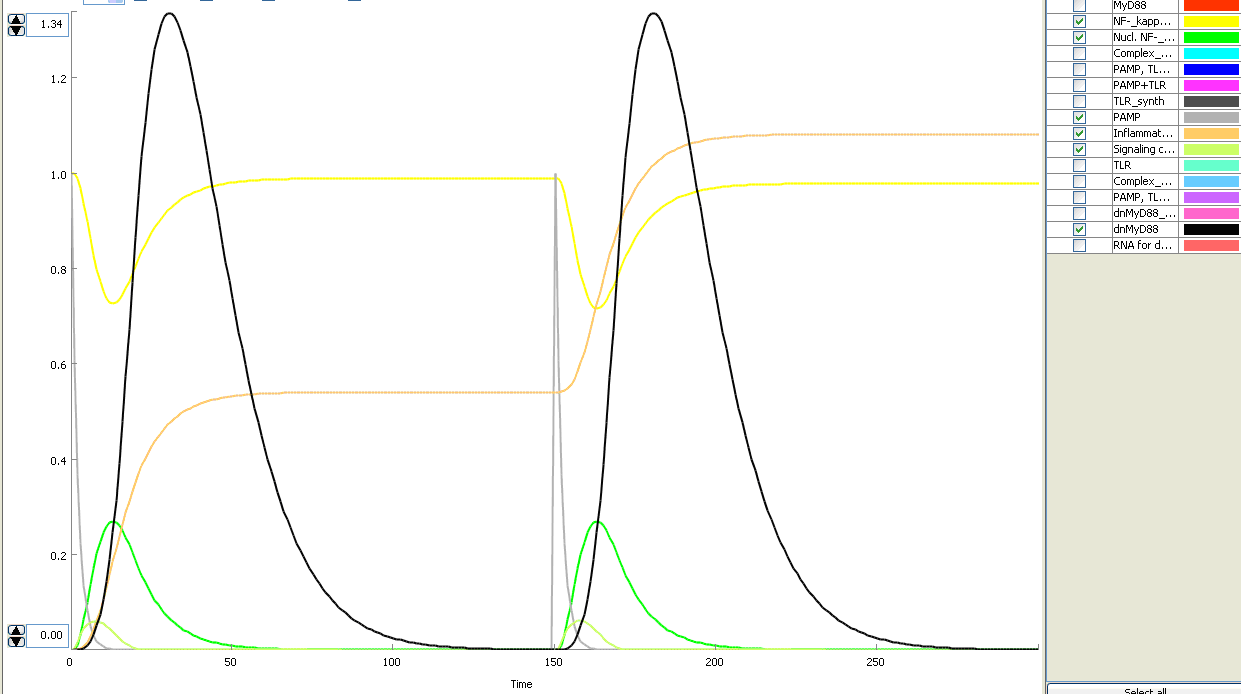
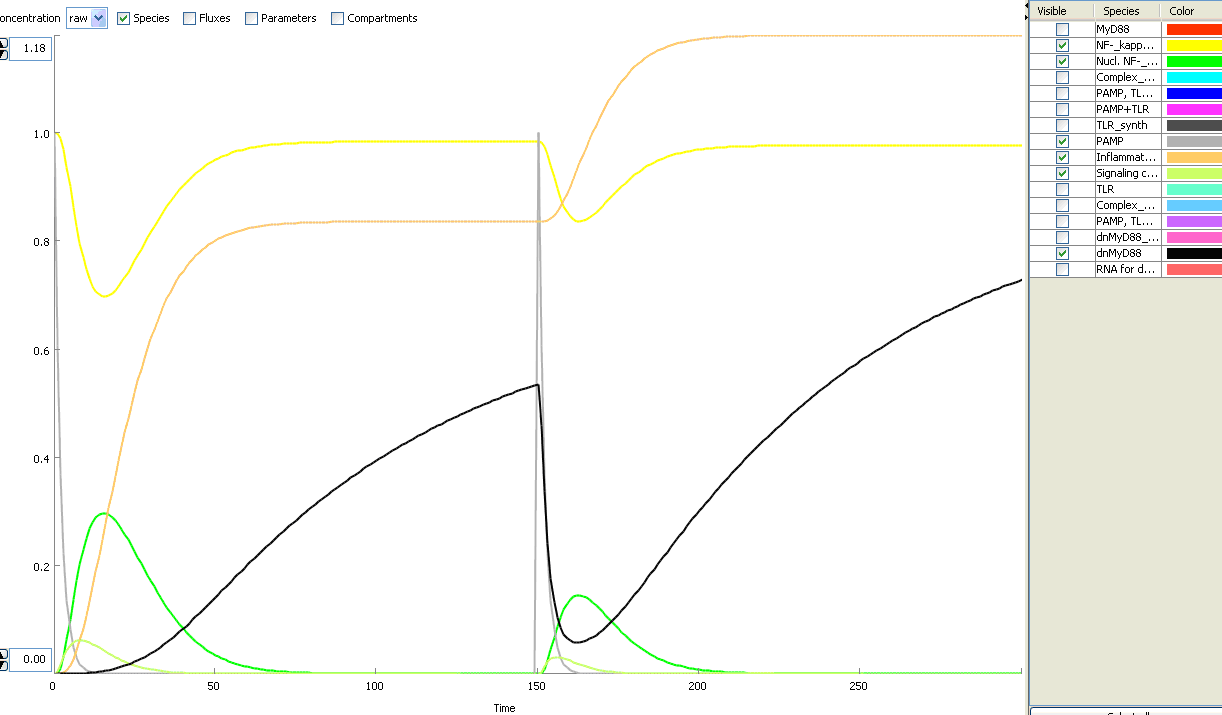
This idea is similar to the natural mechanism of tolerance, which is already present in living cells and which decrease the response to repeated bacterial stimulation. This natural tolerance is activated slowly, on the order of days and operates through several different mechanisms (Figure). Our feedback mechanism (i.e. artificial tolerance) should decrease the response within hours and thus "attack" the signaling pathway at the point, which has not been used in the natural system.
Approach
Model of signaling
Parts design
At first we had to design primers to replicate a desired DNA fragment. In primers we included restriction sites - on left site XbaI and on the right site SpeI, NcoI and PstI. We cloned that part in to BioBrick plasmids with ccdB domain to get all restriction sites needed for BioBrick assembly. We had to design all parts de novo, since no parts like promoters, terminators, desired proteins for signaling pathway modification, degradation flags and reporters had been designed so far - neither to work in mammalian cells. List of desired constructs is shown below. For our use we designed a special vector ([http://partsregistry.org/Part:BBa_J52017 BBa_J52017]) with terminator to simplify constructs assembly. All our composite parts (promoter plus part) were then cloned in this vector.
We also needed fusion proteins e.g. dnMyd88-rLuc-PEST ([http://partsregistry.org/Part:BBa_J52013 BBa_J52013]) - that is our dominant negative protein linked with reporter and degradation flag. This parts are designed like basic parts - not composite, although they are fusion proteins. Between proteins there is a 6 aminoacid long linker because only 2 aminoacid long linker formed during biobrick assembly could affect protein folding. We introduced a six amino acids long linker in between protein - reporter and reporter - degradation flag with primers using PCR Overlap Extension method. These parts were then combined with promoter (NFκB) in BioBrick assembly technique. The part was then inserted in vector with terminator and ready to use in human cells (HEK 293).
| Registration number | Part's Name | Vector |
|---|---|---|
| [http://partsregistry.org/Part:BBa_J52008 BBa_J52008] | rluc | pSB1AK3 |
| [http://partsregistry.org/Part:BBa_J52010 BBa_J52010] | NFκB | pSB1AK3 |
| [http://partsregistry.org/Part:BBa_J52011 BBa_J52011] | dnMyD88-likn-rLuc | pSB1AK3 |
| [http://partsregistry.org/Part:BBa_J52012 BBa_J52012] | rluc-link-PEST191 | pSB1AK3 |
| [http://partsregistry.org/Part:BBa_J52013 BBa_J52013] | dnMyD88-link-rluc-link-pest191 | pSB1AK3 |
| [http://partsregistry.org/Part:BBa_J52014 BBa_J52014] | NFκB+dnMyD88-link-rLuc | pSB1AK3+TER |
| [http://partsregistry.org/Part:BBa_J52016 BBa_J52016] | eukaryotic terminator | pSB1AK3+TER |
| [http://partsregistry.org/Part:BBa_J52017 BBa_J52017] | eukaryotic terminator vector | pSB1AK3 |
| [http://partsregistry.org/Part:BBa_J52018 BBa_J52018] | NFκB+rLuc | pSB1AC3 |
| [http://partsregistry.org/Part:BBa_J52019 BBa_J52019] | dnTRAF6 | pSB1AK3+TER |
| [http://partsregistry.org/Part:BBa_J52021 BBa_J52021] | dnTRAF6-link-GFP | pSB1AK3+TER |
| [http://partsregistry.org/Part:BBa_J52022 BBa_J52022] | NFκB+dnTRAF6-link-GFP | pSB1AK3 |
| [http://partsregistry.org/Part:BBa_J52023 BBa_J52023] | NFκB+rLuc-link-PEST191 | pSB1AK3 |
| [http://partsregistry.org/Part:BBa_J52024 BBa_J52024] | NFκB+dnMyD88-link-rLuc-link-PEST191 | pSB1AK3+TER |
| [http://partsregistry.org/Part:BBa_J52026 BBa_J52026] | dnMyD88-link-GFP | pSB1AK3+TER |
| [http://partsregistry.org/Part:BBa_J52027 BBa_J52027] | NFκB+dnMyD88-link-GFP | pSB1AK3 |
| [http://partsregistry.org/Part:BBa_J52028 BBa_J52028] | GFP-PEST191 | pSB1AK3 |
| [http://partsregistry.org/Part:BBa_J52029 BBa_J52029] | NFκB+GFP-PEST191 | pSB1AK3 |
| [http://partsregistry.org/Part:BBa_J52034 BBa_J52034] | CMV | pSB1AK3+TER |
| [http://partsregistry.org/Part:BBa_J52035 BBa_J52035] | dnMyD88 | pSB1AK3+TER |
| [http://partsregistry.org/Part:BBa_J52036 BBa_J52036] | NFκB+dnMyD88 | pSB1AK3+TER |
| [http://partsregistry.org/Part:BBa_J52038 BBa_J52038] | CMV-rLuc | pSB1AK3+TER |
| [http://partsregistry.org/Part:BBa_J52039 BBa_J52039] | CMV+rLuc-link-PEST191 | pSB1A2 |
| [http://partsregistry.org/Part:BBa_J52040 BBa_J52040] | CMV+GFP-PEST191 | pSB1AK3 |
| [http://partsregistry.org/Part:BBa_J52642 BBa_J52642] | GFP | pSB1AK3+TER |
| [http://partsregistry.org/Part:BBa_J52648 BBa_J52648] | CMV+GFP | pSB1AK3+TER |
Transfection
In September and October we started to work on transfection of our constructs into human embrional kidney cells (HEK293). and three detection system mentioned below. At first we had to optimize the methods (read articles, test negative and positive controls) and learn how to work with human cells. Experiments are still in progress.
Despite transfection with our construct, we also have to transfect cellc with TLRs, because strain HEK293 expresses only TLR3 and TLR6. Sepsis is usually response to pathogenic Gramm negative bacteria. Their outer membrane contains LPS (lipoproteins) that is recognized by TLR4 and accessory molecule MD2. At the beginning we transformed all cells with our constructs and additional plasmids, one coding TLR4 receptor and another MD2. Very soon we found out that our plasmids were contaminated with LPS of E. coli (strain DH5α used for transformation), since there was no difference in response between stimulated (with LPS) and unstimulated cells. To overcome this problem, now we are using TLR5 receptor. This receptor detects bacterial flagelin. Signal transfer through this receptor does not depend on presence of LPS.
Detection systems
We have tested our hypothesis using three different detection systems. All of them had different approach, however we were expecting the same result – synthesis of dominant negative protein (MyD88 or TRAF6), blocking of signalling pathway and consequently transcription termination of dominant negative protein. The construct (parts) we have made are designed for each detection system respectively.
Requests for the optimal detection system were:
- velocity;
- sensitivity;
- paralelization;
- optical signal;
- in vivo detection;
- low price.
On the basis of those requests we have decided for the following detection systems: flow cytometry, luminometry and ELISA. Members of those subgroups incharged for each system are :
Which constuct were used in individual detection system and short description of system itself:
Flow cytometry
Used composite parts: NFκB-MyD-Luc ([http://partsregistry.org/Part:BBa_J52014 BBa_J52014]).
This is a method for very precise detection of each individual cell which is marked. In our case markers were secondary antibodies against phosphorilated ERK kinases. A result of transfection with dnMyD88 or dnMyD88-rLuc and stimulation with flagelin or LPS (depends on chosen TLR receptor) there was activation of signalling pathway and phosphorilation of ERK kinases. After synthesis dominant negative protein should block signalling pathway and amount of phosphorilated kinases should decrease. Further the fluorescence of fluorochromes, conjugated on secondary antibody, would decrease too.
Luminometry
Used composite parts: NFκB-MyD-luc ([http://partsregistry.org/Part:BBa_J52014 BBa_J52014]), NFκB-MyD-luc-PEST([http://partsregistry.org/Part:BBa_J52024 BBa_J52024]), NFκB-luc-PEST([http://partsregistry.org/Part:BBa_J52023 BBa_J52023]), CMV-luc([http://partsregistry.org/Part:BBa_J52038 BBa_J52038]) and CMV-luc-PEST([http://partsregistry.org/Part:BBa_J52039 BBa_J52039]).
With parts that are under NFκB promoter we want to prove inhibitory effect of dnMyD88 on signal pathway (on the basis of luminiscence decrease). Parts with PEST tag should be degradated and therefor luminiscence should increase. We found out that it takes 4 hours for any response.
We also want to calculate half-life of rLuc and rLuc-PEST, so we used those two constructs that are expressed under constituitive promoter [[Troubleshooting, References & Sponsors#Terms|CMV. Synthesis was inhibited by adding cycloheximide in intervals.
ELISA
Used composite parts: NFκB–dnMyD88 ([http://partsregistry.org/Part:BBa_J52036 BBa_J52036]) and NFκB–TRAF6–GFP ([http://partsregistry.org/Part:BBa_J52022 BBa_J52022]).
With this detection system we are detecting active NF-kappaB tranctiption factor. Active form of this protein is present after activation of signalling pathway when protein is released from complex of inhibitors (IKKα, IKKβ, IKKγ). It migrates into nucleus where acts as transcription activator. We are using high bind 96 well plates coated with streptavidine (we prepare it by ourselfes). The biotinilated probe has double stranded NFκB binding sequence and single stranded linkage with a plate. The NFκB from whole cell lysate is captured by a probe. We detect it with primary mouse antibodys and secondary peroxidase-conjugated rabbit anti-mouse antibodies.
Komentarji:
- Časi ... pišemo, kot da še delamo al v pretekliku. Jest sm pisala, kot da še, samo se mi ne zdi primerno oz. bi na konc (predn gremo) mogl spremenit.
- Beseda signalling: kaj je bolj prav. V literaturi se pojavlja t enim ali dvema L-jem. Word smatra za pravilno besedo z enim L.
- Mogoče bi se pri luminomertiji ne pisalo tako podrobno ... recimo funkcije cikloheksamida še jest ne vem natancno, večina bralcev wikija verjetno še manj. Če pa je to bistvenega pomena, pa naj se bolj natančno opiše, da ima nek kontekst.
- označi, kaj pomeni kateri part
- pri slikah vpiši še reference!
- velikost slik nastavi tako, da se bo sploh dalo kaj videti - nihče ne bo klikal na množico majhnih slik in čakal na prenos (odgovarja Rok: Uporabil sem ukaz za prikazovanje slik v galeriji, ker sem tako lahko slike še najbolj smiselno uredil. bom pa poskusil še drugače)
- bolj strukturiraj prvo stran z več linki
- celotni širino strani je težko brati - razdeli v kolone ali ilustriraj tekst s slikami na desni strani
- ali je na voljo kakšen software za lažje oblikovanje wikija ? morebiti lahko vprašamo kakšno drugo ekipi ali Randya kaj so uporabljali.(odgovarja Rok: Nisem zasledil nobenega programa za oblikovanje wikija. V rubriki Recent changes sem zasledli, da vse ekipe, ki aktivno spreminjajo svoje strani, uporabljajo ukaze za wiki, ki so prikazani na strani Help)
| Home | Background and Signalling Pathway | Anticipated Results & Significance | Troubleshooting, References & Sponsors | Team members |
|---|