UT Austin 2005
From 2006.igem.org
The UT Austin team, like last year, is extremely informal. We have no classes nor particularly organized meetings. Anyone who wishes to contribute is welcome to do so. Thanks to the natural enthusiasm of our team and occasional whip cracking from Andy we have made some interesting progress.
Last year we planned on building a light activated edge detector. Thanks to the the incredible contribution of an engineered light detector from Anselm Levskaya at Voigt Lab UCSF we were able to take bacterial photographs (Levskaya et al., Nature, 2005). This was only the first step in our planned goal and in fact we are still working on building the edge detector.
This year we have several project plans which, if they don't self-assemble into green goo and eat the molecular biology building, should be fairly amusing. Being at heart a bunch of hackers, we believe that the greatest contribution to the field will come from actual experiments and thus we are plowing ahead with our experiments while modeling our system.
The UT Austin team is:
- Aaron Chevalier
- Dan Blick
- Eric Davidson
- Jeff Tabor
- Laura Lavery
- Matt Levy
- Rachel Haurwitz
- Zack Booth Simpson
Advisors:
Contents |
Light Wires
We are looking to build circuits which demonstrate the unique regulatory capabilites of the light dependent system developed by the UT/UCSF team last year. One such instantiation is the original Edge Detector (see below) from last year which we are finally finishing.
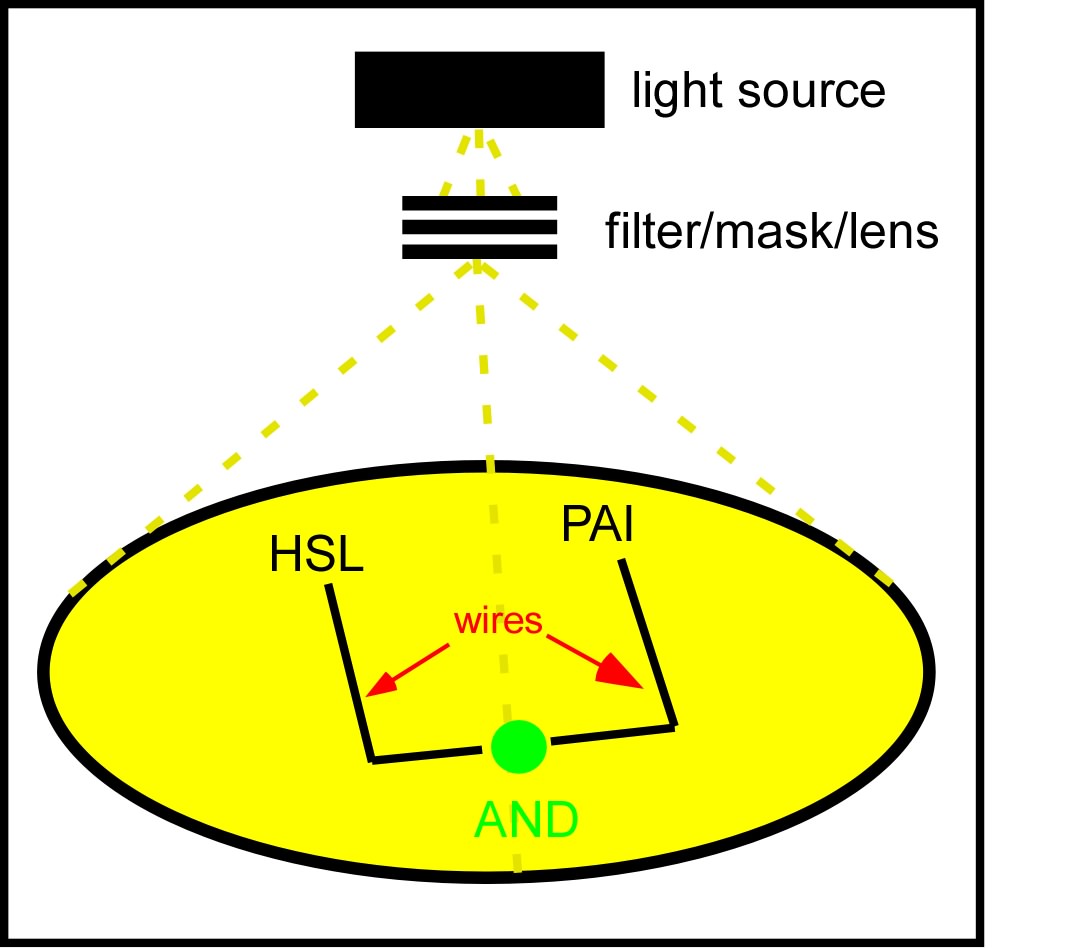
The idea which came out on top of this year's heap was to transform a genetically identical lawn of E.coli into a biochemical circuit board, using light to dictate the conductive potential of each ~1x3 micron segment of surface area. All cells on the lawn will carry two biochemical signal amplifiers: a 3OC6HSL amplifier I15030, and a pseudomonas autoinducer (AI-1) amplifier J13030. These signal amplifiers will be inactive in certain locations of the plate and active in others. We will dictate which E.coli on the plate have active circuits with light, by using J13100 to strongly repress these two signal amplifiers in 660nm light.
We will use light to draw intersecting wires on the E.coli canvas, and will activate biochemical signal propagation down each wire with a separate quorum sensing signal (see figure). The exogenous addition of a signal, say 3OC6HSL will activate high level production of that compound in the cells into which it diffuses. The newly produced 3OC6HSL will diffuse out of the biochemically active cells in all directions. Those cells which neighbor the activated cells AND which are in the light will similarly be triggered to actively produce the 3OC6HSL compound. This process will continue down the region of the agar plate, moving from cell to cell as far as the light allows.
We will propagate orthogonal biochemical currents down separate wires (figure), and will genetically encode different logical operations which will be relevant at the intersection of the two wires. The simplest logical operation will be an AND gate. If two orthogonal signals succesfully propagate down the plate and intersect at the end, two complementary halves of a reporter gene (LacZ) will be expressed and become functional, signaling an output of 1 from the AND gate.
Model
To model the light wires system, we constructed a stochastic simulation of gene regulation in the cells based on equilibium constants and kinetic rates from the literature. We believe that stochastic fluctuations will be important in our system because of (1) the small number of relevant molecules present in each cell and (2) feedback effects. Our approach was inspired by the Arkin model of phage lambda [1]. The simulation implements the original Gillespie algorithm [2] and uses a partition-function based model of gene repression due to Shea and Ackers [3].
Using a simple model of diffusion, we predicted that leaky expression of LuxI would be enough to start the feedback circuit and turn some 'off' cells 'on'. This supported a decision to add AiiA to the circuit to help clear up excess HSL.
[1] Arkin A, Ross J, McAdams HH. Stochastic kinetic analysis of developmental pathway bifurcation in phage lambda-infected Escherichia coli cells. Genetics. 1998 Aug; 149(4):1633-48.
[2] Gillespie DT. Exact stochastic simulation of coupled chemical reactions. J. Phys. Chem, 1977.
[3] Shea MA, Ackers GK. The OR control system of bacteriophage lambda. A physical-chemical model for gene regulation. J Mol Biol. 1985 Jan 20;181(2):211-30.
Edge Detector
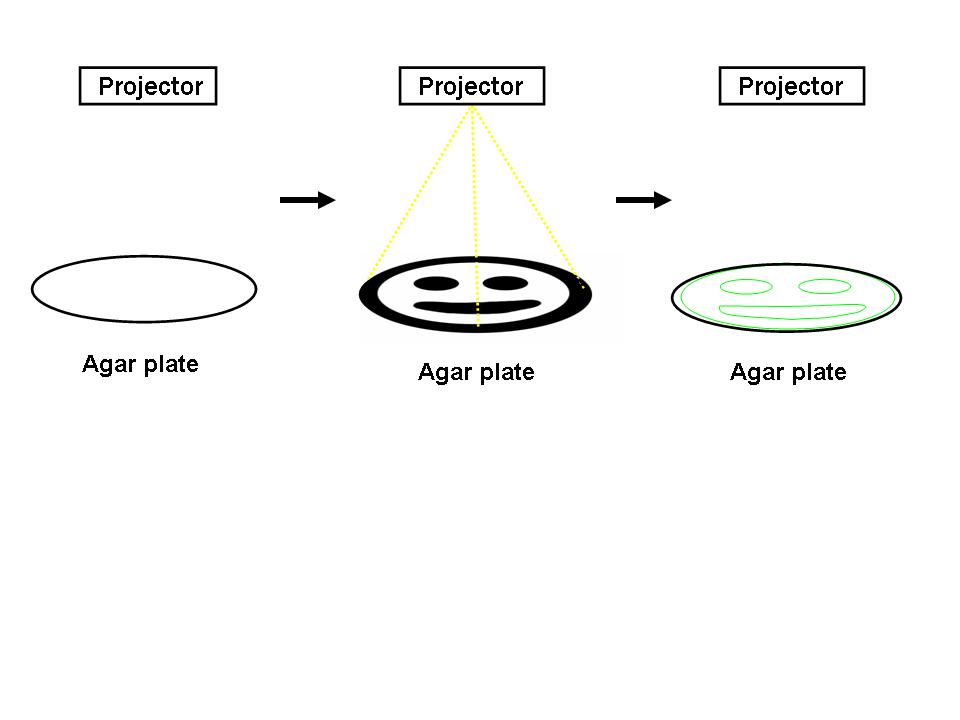
For the 2004 Synthetic Biology Competition, our joint UT-Austin/UCSF team designed a genetically encoded edge detection device. In our system, a lawn of genetically identical precursor E.coli would be subjected to a light image, and only those cells at the light/dark boundary would become active and express a genetic reporter.
An obvious hurdle in the implementation of this system was genetically encoding light detection in an easily manipulate-able and tractable system like E.coli. To accomplish this, we used an incredible part engineered in the Voigt lab. This part, Cph8, (I15010) is an engineered fusion between a cyanobacterial light sensing phytochrome (Cph1) and an E.coli transmembrane histidine kinase, (EnvZ). 660nm light causes an isomerization in the Cph1 domain of the chimera which strongly inactivates the histidine kinase acitity of EnvZ. When EnvZ is inhibited, a phosphorelay cascade which activates transcription from the OmpC promoter R0082) and inhibits transcription from the OmpF promoter (R0084). We showed that when this system is expressed in E.coli, it is possible to transform each cell on an agar surface into a decision making pixel capable of deciding whether it is in the light or dark. The community of cells is therefore capable of genetically reproducing a light image (figure to come).
This was the first step in engineering the edge detector, a massively parallel computation system that should be able to easily compute the edge of a complex light image; a very hard serial computation problem. We have now built the entire edge detection circuit and are currently testing/optimizing.
Comment on the format of the Jamboree as it increases in size -JT, 11/8/2005
Some have proposed that the quality of a team's wiki page should be the determining factor in deciding which teams present orally at future conferences. The teams not chosen to present based on their wiki would give posters.
I think documenting projects is critical. I think wiki's are a good way to do that. I think teams should be encouraged (or even required) to have wikis in the interest of the field and the Jamboree.
I don't think wiki quality should define the quality of the work. The competition is about promoting creative thought and training young synthetic biologists. As far as choosing talks, maybe an abstract would suffice? For example, our team never would have been able to present (this year or last) if this rule were in place. However, I think we've done work that has sparked interest in the field of synthetic biology and the jamboree.