Brown:Bacterial Freeze Tag
From 2006.igem.org
Contents |
Bacterial Freeze Tag
Overview
This project involves programming bacteria to be able to play a game of freeze tag. Bacteria will be engineered to swim around a microfluidics device until they reach a certain proximity to the 'IT' cell and then they will lose their ability to move. This loss of motility will be combined with a change in color from Green to Blue. When another bacterium, which is moving (not the 'IT' cell), reaches a certain proximity to the 'frozen' bacteria it will again regain its ability to move and turn from Blue to Yellow.
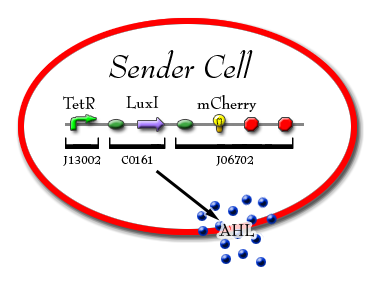
Sender Cell
TetR promoted with LuxI downstream. LuxI is an enzyme that produces AHL and will produce the red fluorescent protein (RFP). The AHL produced is exported from the cell where it then forms a complex with the LuxR protein that is produced by the AHL sensor within the Receiver cell.
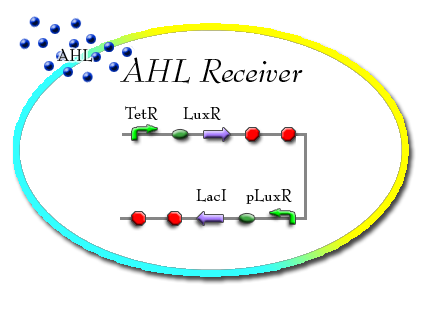
AHL Sensor
The AHL sensor is TetR promoted and forms the LuxR protein which then forms a complex with AHL. This LuxR and AHL complex then activates the pLuxR promoter. Downstream of the pLuxR promoter is the LacI protein. LacI inhibits the pLac promoter on the "Freeze Machine".
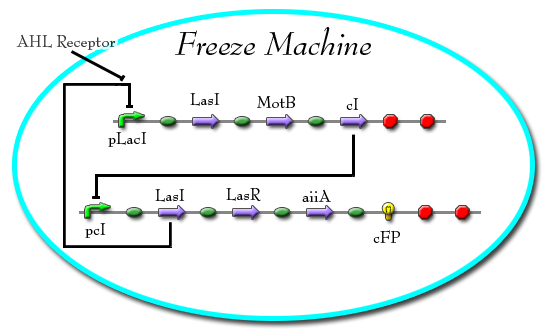
'Freeze Machine'
A promoter that is regulated by LacI will promote the production of LasI, MotB, and cI. This will subsequently inhibit the production of CFP and LasR. In the presence of LacI, however, MotB, LasI, and cI will not be produced. CFP will therefore be produced along with LasR and LacI. This results in the "freezing" of the cell.
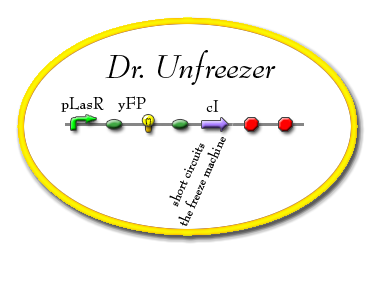
"Dr. Unfreeze"
"Dr. Unfreeze" is responsible for short circuiting the freeze machine. When it is activated by AI-1 and LasR it begins producing cI, which will inhibit pcI and allow the cell to begin producing MotB.
What we achieved
One of the major issues concerning creating our game of bacterial freeze tag was that we were unsure if a single bacterium ("IT" cell) would be able to send a sufficient AHL signal. In their native environment, bacteria use molecules like AHL as quorum-sensing molecules and therefore the response is based on many bacteria releasing the signalling molecule. In our system, however, we depend on the ability of a single bacterium to be able to communicate with this same signalling molecule. Since we will be using a strong promoter to drive expression of AHL, it is possible that this will be feasible.
To test this, we designed a plan to mix bacterial sender cells (AHL producers) with bacterial receiver cells (AHL receivers) with downstream reporters. In this way, we could easily measure the communication between the senders and the receivers by measuring fluorescent signal. We could experiment with various concentrations of sender cells and determine our key question of whether a single bacterial cell could elicit a response.
Challenges
During the process of building this system, several road blocks were presented that caused the team to put a hold on the project. Although we were successful at creating the AHL receiver component of the receiver cell, the AHL sender cell, provided much frustration. We were unable to get the AHL sender to emit its proper red flouresence even though sequecing showed proper ligations of the parts. Also, while we were working away at this system, we realized that much research was needed into developing a microfluidics chamber, (the arena, if you will), where the engineered bacteria would be able to survive and be active in this complex game of freeze tag.
Future work
There are plans of developping a proper microfluidics chamber in which the bacteria will be able to be active. Also, in the coming months we intend on further investigating why the AHL sender cell failed to emit the red fluoresence. Once the AHL sender cell is functioning, we will determine how much AHL is emitted from an individual sender cell. Such quanitification will be necessary in order to figure out how many sender cells will be necessary to emit enough AHL to stop the motility of a nearby bacterium.